
High-resolution, non-invasive OCT imaging that reveals
skin structure and microvasculature in 3D — in just seconds.
15-second scan • >1 mm imaging depth • 6 × 6 mm field of view • real-time 3D rendering
VivoSight gives you
The Whole Picture®
VivoSight provides fast, non-invasive visualisation of skin structure and microvasculature, supporting confident assessment in both clinical practice and skin research.
-
Confident clinical assessment
VivoSight’s unrivalled >1 mm imaging depth and 6 × 6 mm field of view deliver full visualisation of lesion architecture, not partial sampling — to aid clinicians in their assessment of a patient’s clinical conditions
-
Efficient
workflowsA complete, high-resolution 3D scan is
acquired in just 15 seconds, capturing
the entire lesion in a single non-invasive
scan. -
Better patient and subject experience
Completely non-invasive, scar- and pain-
free imaging will delight patients and enable
repeat assessment over time.
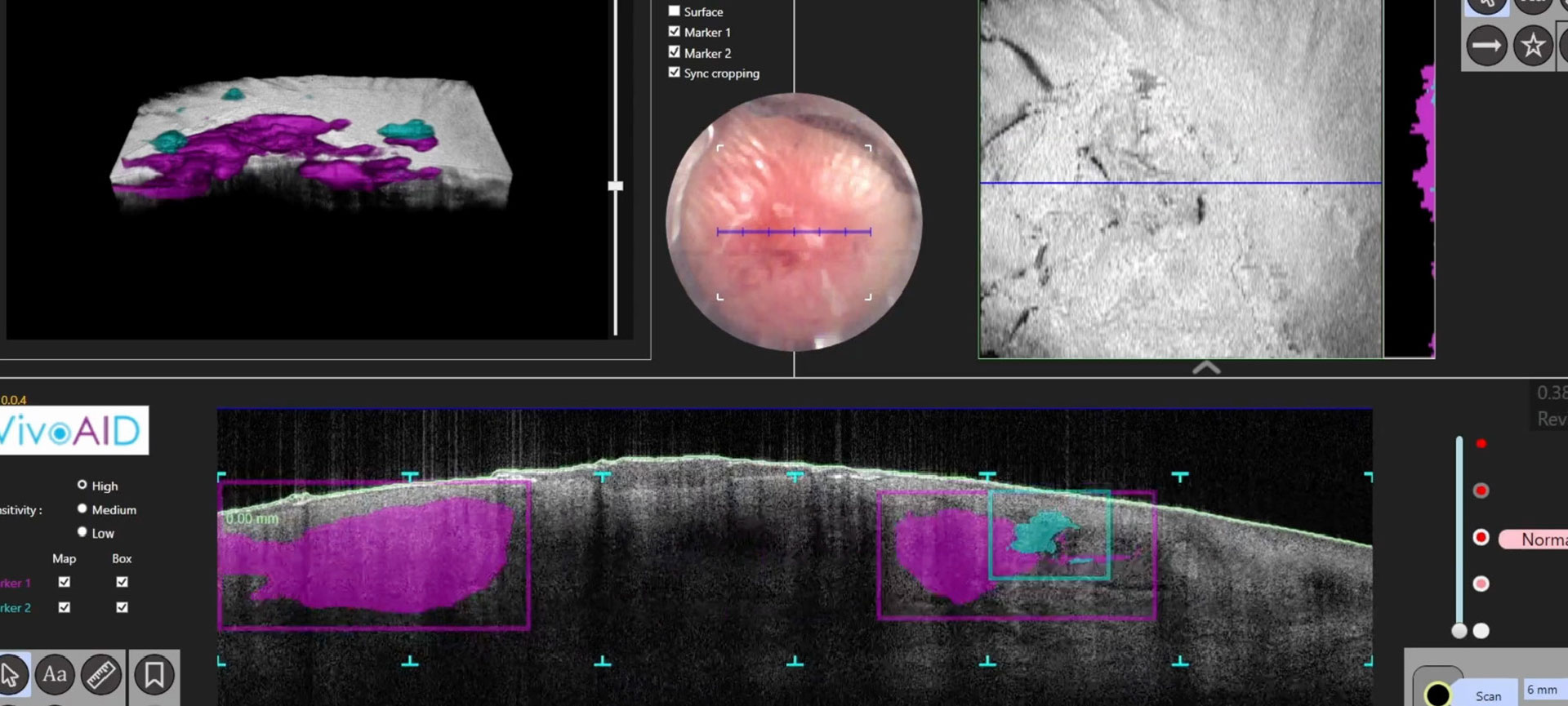
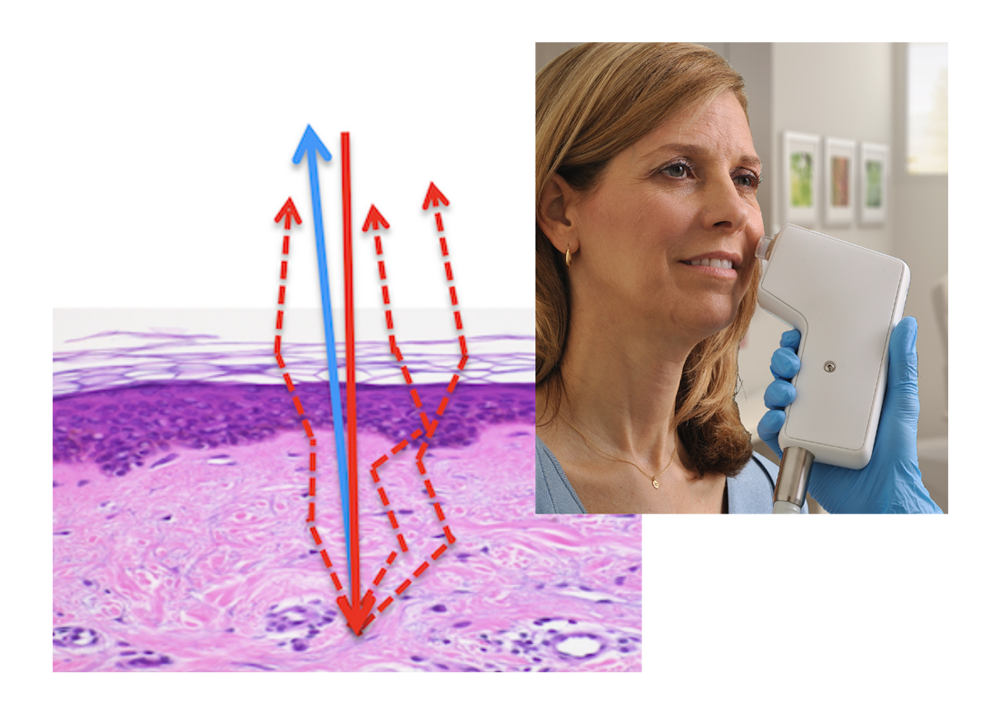
How OCT skin
imaging works
Optical Coherence Tomography (OCT) is an optical analogue of ultrasound imaging.
Using eye-safe infrared light, VivoSight captures a high-resolution 3D block of image data, revealing skin structure and function beneath the surface. This enables real-time, in-clinic visualisation of skin architecture without cutting, scarring, or waiting for histology.

Interested? Schedule a Demo Today!
Our team is available for your online demo of VivoSight skin imaging. Complete the form in the link below to schedule a time with one of our experts.

Applications across dermatology and skin research
-
Skin Research
Enable non-invasive, repeatable imaging for academic and commercial skin health research.
Trusted by clinicians and
researchers worldwide
Makes OCT skin imaging accessible, faster,
and easier to use.
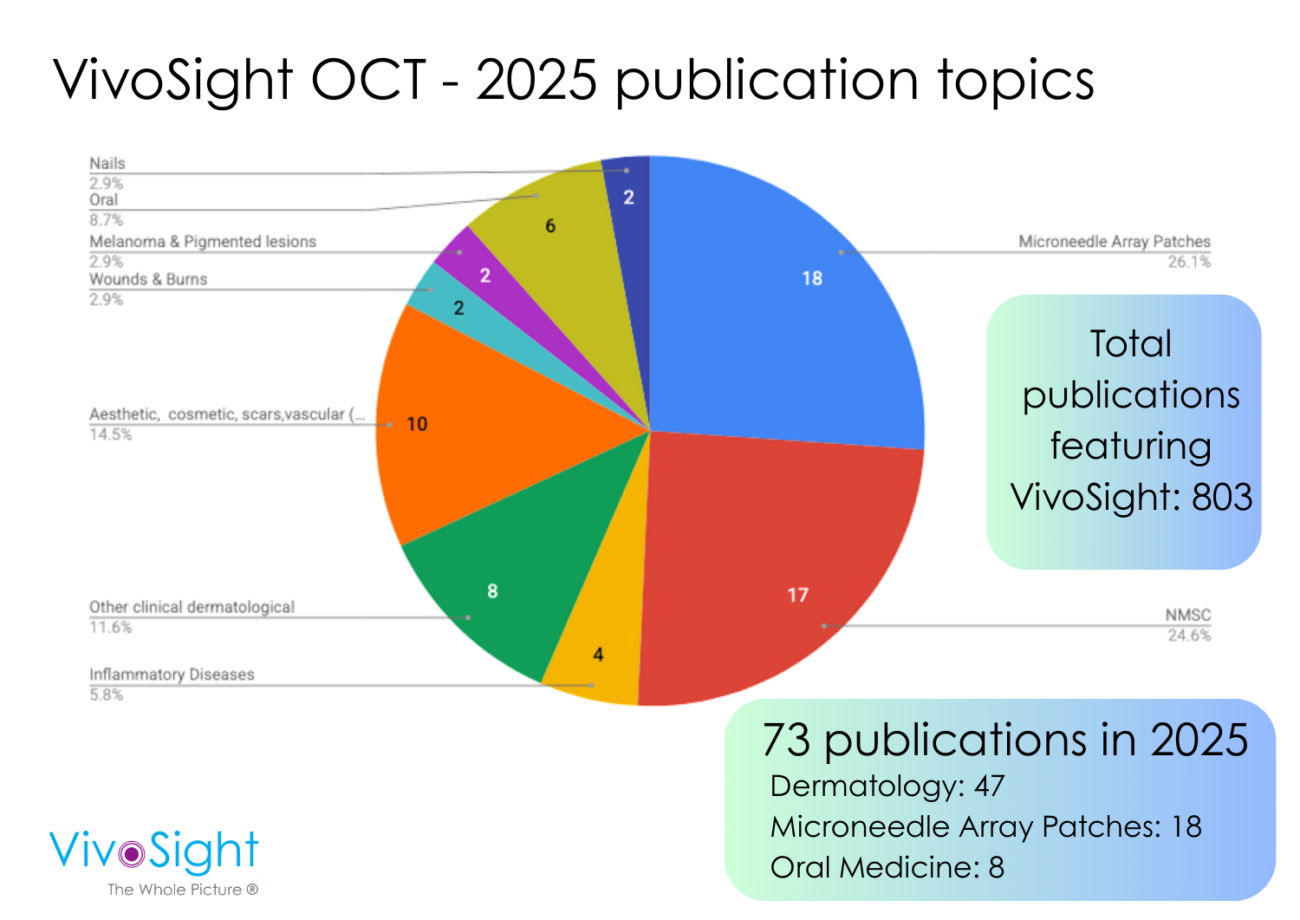
- 800+ peer-reviewed publications
- 200,000+ patient scans completed
- Used routinely in hospitals, clinics and research labs globally
- FDA Cleared
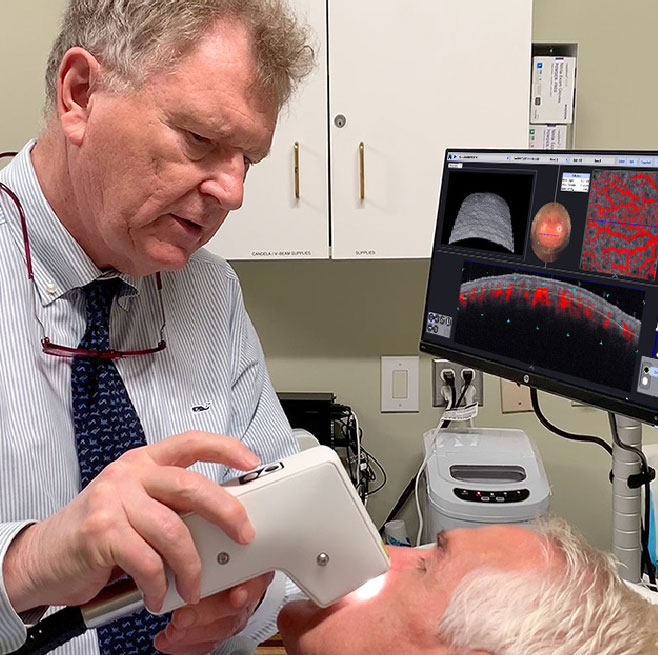
Prof. C. Zachary, UCI, using VivoSight to examine a patient
News & Insights

See VivoSight in Practice
Tailored to your clinical or research needs
Book an online demo with our team to see how VivoSight can support your clinical or research workflow.




